HOWTO: Use beads to correct for day-to-day variation¶
Flow cytometers are complex instruments, and the precise numerical values they report depend not only on the fluorescence of the sample, but also on the illumination intensity, optical setup, gain of the detectors (ie PMT voltages), and even things such as whether the flow cell has been cleaned recently. We often want to compare data that was collected on different days, and this instrument drift can make these comparisons difficult.
Many cytometers, especially those used in a clinical setting, use a daily calibration to counter these effects. However, these calibrations are intended for standardized protocols that are run regularly, not the one-off experiments that many investigators run.
One approach to this problem is to calibrate each day’s measurements using a stable calibrant. The idea is straightforward: each day, in addition to your experimental samples, you measure a sample of stable fluorescent particles (of known fluorescence), then use these calibrant measurements to convert the arbitrary units (au) for your experimental samples to the known units for your calibrant. For example, if your particles have a fluorescence of 1000 molecules of fluoresceine (MEFL), and you measure their brightness to be 5000 au, then you know that an experimental sample with a brightness of 10,000 au is equivalent to 2,000 MESF. This relationship holds even if tomorrow the laser is a little dimmer or the fluidics are a little dirty.
While there are a number of different kinds of stable calibrants that might be
used this way, we have had very good success with
Spherotech’s Rainbow Calibration Particles.
(We usually refer to these particles as “beads”.) Because they’re made of
polystyrine with the fluorophores “baked in” (not on the surface), they
are ridiculously stable. Spherotech also provides enough technical data
about their fluorescence to make it easy to determine a calibration curve
and apply it to experimental data. Below, you can find the process for
doing so using Cytoflow.
Note
It is often considered “best practice” only to compare data that was on the same instrument with the same detector gain settings. Using beads this way can help you get around the “same detector gain settings” requirement – for example, if you have a really bright sample and a really dim sample – but it is not a good idea to try to compare between different instruments unless those instruments have exactly the same optical configuration (lasers, filters, and from the same vendor.)
Procedure¶
Collect a sample of beads on the same day, and with the same settings, as your experimental samples.
Import your data into
Cytoflow. Do not import your bead control. In the example below, we’ll have three fluorescence channels – Pacific Blue-A, FITC-A and PE-Tx-Red-YG-A – in addition to the forward and side-scatter channels.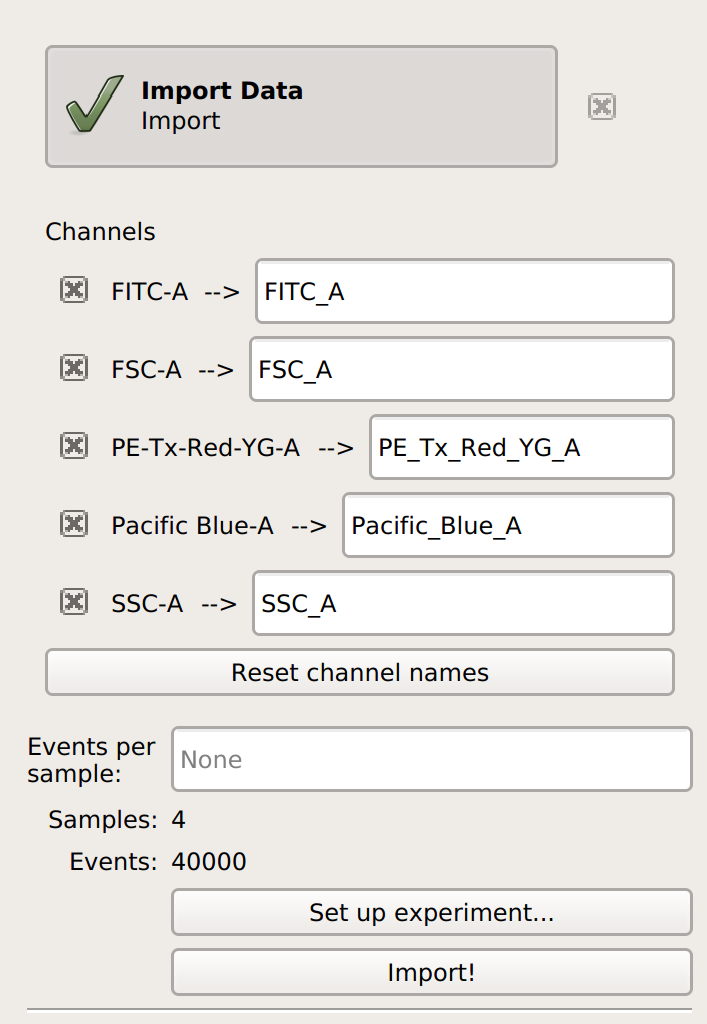
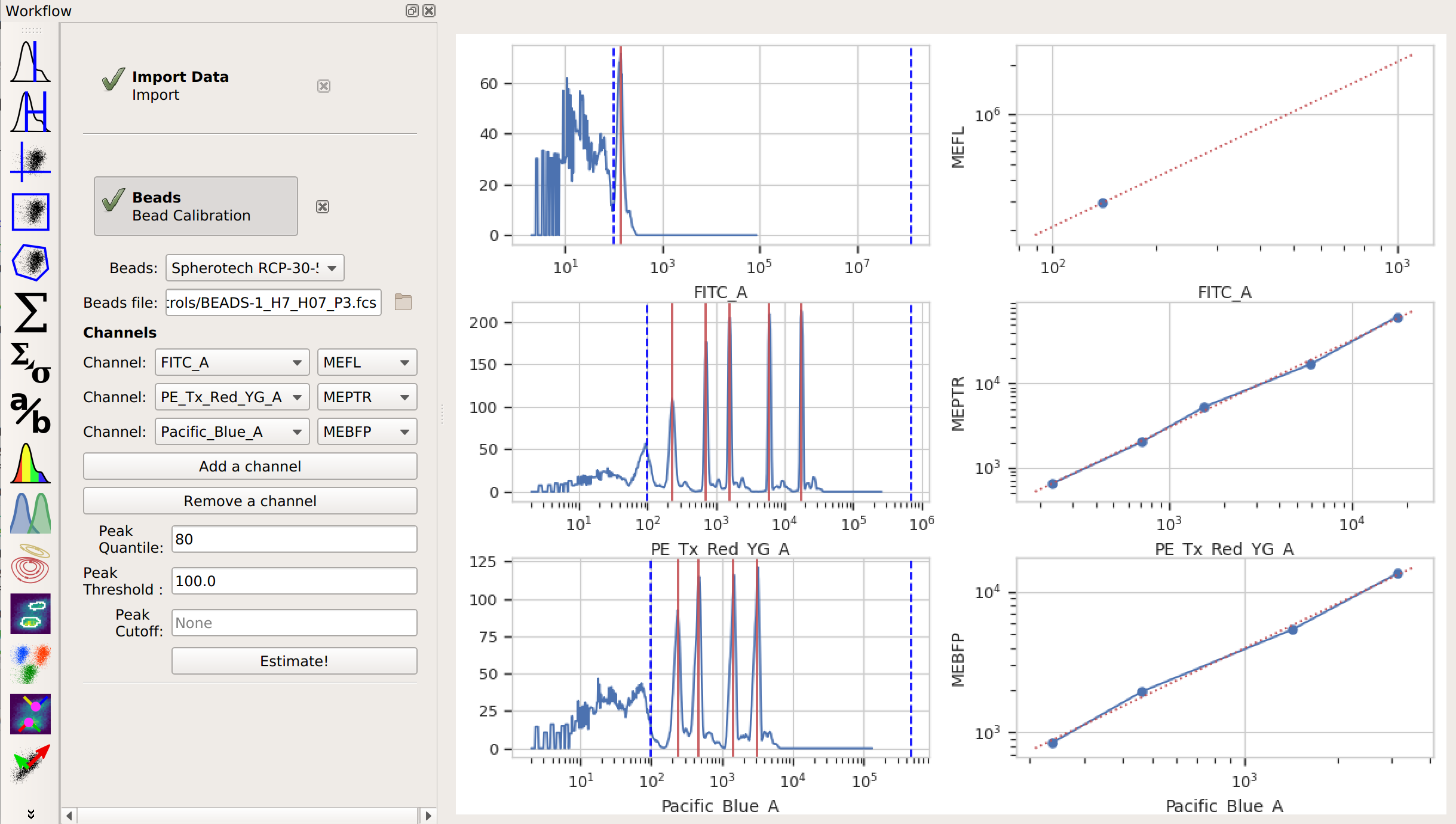
Add the Beads operation to your workflow. It’s the
 icon.
icon.Choose the beads you used, including the specific lot, from the drop-down list.
Note
If the beads you want to use are not in the list, please submit a bug report. Adding a new set of beads is pretty trivial.
Specify the file containing the data from the beads.
Click Add a channel for every channel you want to calibrate. In the Channels list, specify both the channel you want to calibrate and the units you want to calibrate it.
Note
It works best to choose units of a fluorophore that is spectrally matched to the channel that you’re calibrating. Here are the beads that
Cytoflowknows about (including the laser and filter sets used to characterize the beads):Spherotech ACP 30-2K
Spherotech RCP-30-5A Lot AN04, AN03, AN02, AN01, AM02, AM01, AL01, AK04, AK03 & AK02**
Spherotech RCP-30-5A (Euroflow) Lot EAM02 & EAM01
Sphreotech RCP-30-5A (Euroflow) Lot EAK01, EAG01, EAE01 & EAF01
Spherotech RCP-30-5A Lot AK01, AJ01, AH02, AH01, AF02, AF01, AD04 & AE01
Spherotech RCP-30-5A Lot AG01
Spherotech RCP-30-5A Lot AA01, AA02, AA03, AA04, AB01, AB02, AC01 & GAA01-R
Spherotech RCP-30-5A Lot AC02, AC03 & AD01
Spherotech RCP-30-5A Lot Z02 and Z03
Spherotech RCP-30-5 Lot AA01, AB01, AB02, AC01 & AD01
Spherotech RCP-30-5 Lot AM02, AM01, AL01, AH01, AG01, AF01 & AD03
Spherotech RCP-60-5
Spherotech URCP 38-2K
Spherotech URCP 38-2K Lot AN01, AM01, AL02, AL01, AK03, AK02, AK01, AJ02 & AJ03
Spherotech URCP 50-2K Lot AM01 & AJ01
Spherotech URCP 50-2K
The Spherotech fluorophore labels and the laser / filter sets used to measure them (that I know about) are:
MECSB (Cascade Blue, 405 –> 450/50)
MEBFP (BFP, 405 –> 530/40)
MEFL (Fluroscein, 488 –> 530/40)
MEPE (Phycoerythrin, 488 –> 575/25)
MEPTR (PE-Texas Red, 488 –> 613/20)
MECY (Cy5, 488 –> 680/30)
MEPCY7 (PE-Cy7, 488 –> 750 LP)
MEAP (APC, 633 –> 665/20)
MEAPCY7 (APC-Cy7, 635 –> 750 LP)
Click Estimate! Check the diagnostic plots to make sure that each peak in your data was found, and that you have a fairly linear relationship between the (measured) peaks and the (known) calibration.
Note
If not all of the peaks were identified, try messing around with the peak-finding parameters.
Note
Bead calibration is particularly powerful when combined with the autofluorescence correction and bleedthrough compensation described in HOWTO: Compensate for bleedthrough. They’re so useful when done together that this sequence of operations has its own module – see HOWTO: Use the TASBE workflow for calibrated flow cytometry.